A very important facet of pregnancy is the cross talk between the fetus and the pregnant mother. Mouse knockouts are a great model to study the function of genes in the development and maintenance of the organism, however they have a limitations in that the fetal genome cannot be manipulated separately from the maternal genome.
The purpose of this study is to produce a strain of animal that will facilitate the understanding of fetal maternal signaling. A key to the strategy is to specifically knock genes out in the precursors to sperms and eggs i.e. the primordial germ cells (PGC’S). To do this we need firstly to make a transgene that will express an inducible Cre Recombinase. The mouse ddx4 (ddx4) promoter that will be hitched to Cre Recombinase (ddx4-iCreT2) to allow its production specifically in the PGCs. iCreT2 (Chambon et al.) is an inducible Cre recombinase that is a fused to a mutated estrogen receptor that binds tamoxifen and in its presence is translocated to the nucleus where it can function to recombine the specific DNA sequences (lox sites) that are inserted in the introns of specific gene allele knockins thereby disabling a particular gene only when tamoxifen is present. Injection of pregnant animals with tamoxifen that have two copies of a loxed gene of interest and the PGC specific transgene driving Cre Recombinase will cause recombination of the gene of interest in the PGC’s of the pups. These pups will have mutated genes in their germline (eggs and sperm). When these pups mature they are mated to produce the next generation of fetuses with completely mutated copies of the genes in all cells while the mother remains somatically normal. i.e. all cells other than germ cells are normal. In this way we will be able to understand the contribution of a genes function fetally for the maintenance of pregnancy while separating it from the maternal gene function.In most cases genes that are of interest to fetal development have reproductive effects on the adult and thus knockout mothers are not viable which makes it impossible to create a complete litter of knockout pups, i.e. 100 % knockout pups. Even in cases where we can produce knockout pups and where genes allow the knockout parents to be reproductively viable we cannot separate the maternal and fetal compartment i.e. study the fetal effects in a vacuum.
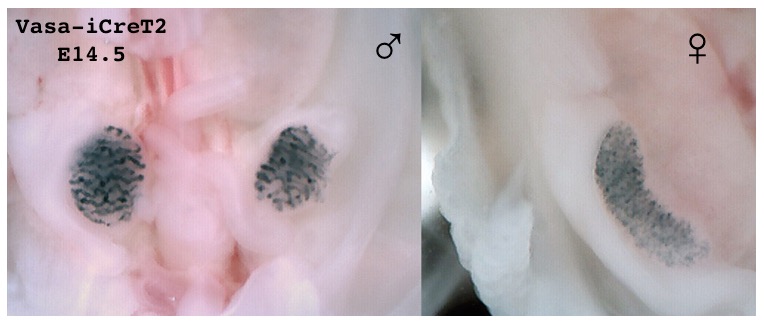
Figure: LacZ expression (Rosa-LacZ Reporter mouse) after tamoxifen induction of E14.5 embryo with vasa CreT2 BAC transgene. This strains allows us to inducibally knock out out loxed genes in sperms and eggs allowing for a fetal specific knockout.